Specimens for dna barcoding
COLLECT SPECIMENS AND RECORD SPECIMEN COLLECTION INFORMATION
a. For beginners to insect collecting the Florida Bug Club is a good place to find information.
b. For those eager to expand to large-scale regional inventories, Janzen et al. (2009) provide details of a mammoth “guinea pig” DNA barcoding initiative in Area de Conservacion Guanacaste, Costa Rica.
c. When collecting insects for DNA barcoding, collectors should consider the data required by BOLD and GenBank to make sequence records “BARCODE” compliant.
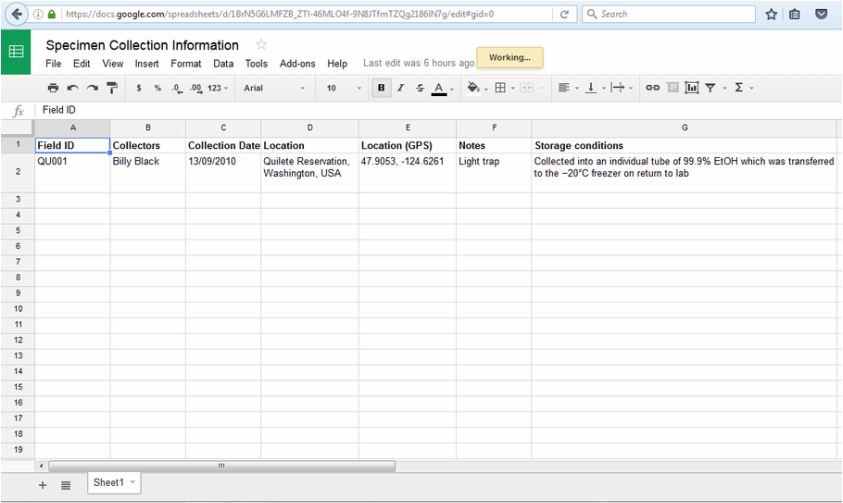
d. The collector should note the way in which the insect was collected, e.g., light trap.
e. The storage conditions should be recorded—Was the specimen stored in ethanol (EtOH)? Was it oven-dried? This information is crucial for understanding incidents of low DNA yield.
f. Care should be taken to prevent DNA degradation in your collected specimens. We advise specimens be collected into an individual tube of 99.9% EtOH which is later deposited into a freezer (−20°C). If this is not possible, longer term storage under ethanol at room temperature should suffice.
g. The ethanol should be frequently changed to ensure it remains at high concentration (as water diffuses out of the specimen, the ethanol concentration decreases).
h. Museum collections can also be mined for DNA barcoding. The most recently collected specimens and those preserved in ways which minimize DNA degradation are preferred, as are those with a good “record” (i.e., with information available to complete the "Specimen Collection Information" table, and especially types).
i. To find the GPS coordinates of a place on Google Maps, follow the steps below.
Click on the table below, download as an excel file, and complete for your specimens. For Field ID please put your initials followed by the sample number e.g. (JJW)A1, (JJW)A2, so that all workshop participants codes are unique.
Save the excel file as "Specimen Collection Information" on your Desktop.
a. For beginners to insect collecting the Florida Bug Club is a good place to find information.
b. For those eager to expand to large-scale regional inventories, Janzen et al. (2009) provide details of a mammoth “guinea pig” DNA barcoding initiative in Area de Conservacion Guanacaste, Costa Rica.
c. When collecting insects for DNA barcoding, collectors should consider the data required by BOLD and GenBank to make sequence records “BARCODE” compliant.
d. The collector should note the way in which the insect was collected, e.g., light trap.
e. The storage conditions should be recorded—Was the specimen stored in ethanol (EtOH)? Was it oven-dried? This information is crucial for understanding incidents of low DNA yield.
f. Care should be taken to prevent DNA degradation in your collected specimens. We advise specimens be collected into an individual tube of 99.9% EtOH which is later deposited into a freezer (−20°C). If this is not possible, longer term storage under ethanol at room temperature should suffice.
g. The ethanol should be frequently changed to ensure it remains at high concentration (as water diffuses out of the specimen, the ethanol concentration decreases).
h. Museum collections can also be mined for DNA barcoding. The most recently collected specimens and those preserved in ways which minimize DNA degradation are preferred, as are those with a good “record” (i.e., with information available to complete the "Specimen Collection Information" table, and especially types).
i. To find the GPS coordinates of a place on Google Maps, follow the steps below.
- Open Google Maps.
- Right-click the place or area on the map.
- Select "What's here?"
- A card appears at the bottom of the screen with the GPS coordinates.
Click on the table below, download as an excel file, and complete for your specimens. For Field ID please put your initials followed by the sample number e.g. (JJW)A1, (JJW)A2, so that all workshop participants codes are unique.
Save the excel file as "Specimen Collection Information" on your Desktop.
TISSUE SUBSAMPLING
Each sample to be used for DNA extraction needs to be in its own small (0.2ml) tube or well of a 96-well plate.
For large samples only a small piece of tissue needs to be sampled (about a 2–3-mm-long piece of insect leg) for DNA extraction. If necessary, refer to more details on the the tissue subsampling process in section 3.6 of the DNA barcodes for insects book chapter.
For large samples only a small piece of tissue needs to be sampled (about a 2–3-mm-long piece of insect leg) for DNA extraction. If necessary, refer to more details on the the tissue subsampling process in section 3.6 of the DNA barcodes for insects book chapter.